CURRENT LAB OPENINGS AND INFORMATION ON AVAILABLE
RESEARCH PROJECTS
Research SCOPE AND PHILOSOPHY
We solve technological
problems that arise at the boundary between the physical world and computing
machines. We develop intelligent sensory microelectronic systems that are
optimized to solve such problems.
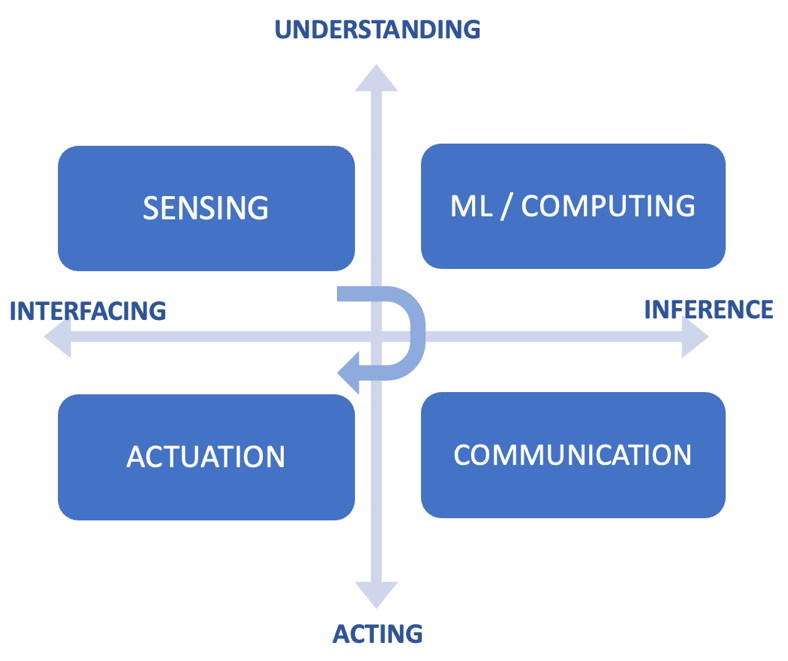
Such systems typically
combine the capabilities of: (1) sensing – for information acquisition, (2)
computing – for machine learning acceleration, (3) communication – for data
readout, and/or (4) actuation – for feedback to the physical world.
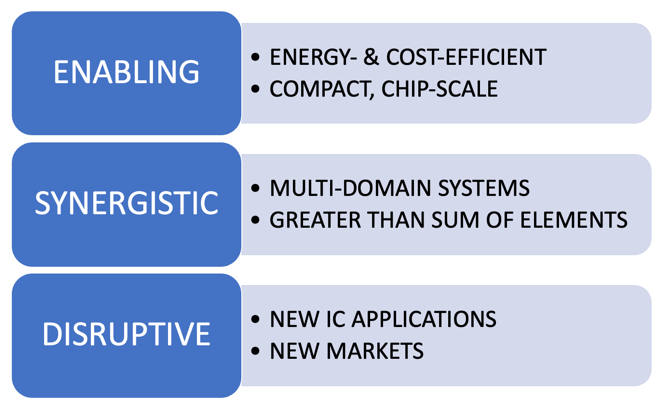
We strive to find
technological solutions that are enabling, synergistic and disruptive.
Research THRUSTS AND GOALS
Currently we focus on the following three key
research thrusts.

Our first goal is to
improve health and wellness by developing effective microelectronic technologies for
neurological monitoring, diagnostics and therapy. Our miniature artificially
intelligent medical devices interface with the nervous system in order to
monitor its function and provide therapeutic feedback. A major current focus of
our research is on responsive neuromodulation of the central and peripheral
nervous systems, aiming to offer effective therapies for intractable epilepsy
and spinal cord injury.

Our second goal is to
enable versatile low-cost computational imaging by developing fast
field-programmable image sensors. This is a fundamentally new class of image
sensors that break the trade-off between the speed and cost of computational imaging
systems, enabling cameras that are both low-cost and high-speed. Their
field-programmable nature yields a vast range of never
seen before capabilities. These cameras bring computational imaging techniques
to a wide range of applications where the scene or illumination change rapidly,
such as autonomous vehicles, drones, industrial robots, augmented reality,
automated surveillance and action cameras.

Our other objectives
include exploring various emerging sensory and computing modalities, such as electrochemical
sensing, computing in memory and nuclear magnetic resonance on a chip.
We always look forward to having talented graduate students from the
University of Toronto and from around the world join our team!
